|
But nitrogen, oxygen, phosphorus, sulfur, halogens, and several other kinds of atoms can also be attached as part of an organic molecule. The most common other element is hydrogen, which makes the family of compounds known as hydrocarbons. The fifth reason is that all of the electrons that are not being used to bond carbon atoms together into chains and rings can be used to form bonds with atoms of several other elements. These different molecules are called isomers. The fourth reason is that the same collection of atoms and bonds, but in a different geometrical arrangement within the molecule, makes a molecule with a different shape and hence different properties. And a molecule that differs by even one atom or one bond position is a molecule of a different compound. This makes for a huge number of possible bond combinations at different places, making a huge number of different possible molecules. The third reason is that carbon atoms can share not only a single electron with another atom to form a single bond, but it can also share two or three electrons, forming a double or triple bond. There is practically no limit to the number or complexity of the branches or the number of rings that can be attached to them, and hence no limit to the number of different molecules that can be formed. They can even join "head-to-tail" to make rings of carbon atoms. 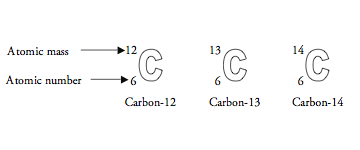
The second reason for carbon's astounding compound-forming ability is that carbon atoms can bind to each other not only in straight chains, but in complex branchings, like the branches of a tree. Silicon (Si), another element in group 14 of the periodic table, also has four valence electrons and can make large molecules called silicones, but its atoms are too large to fit together into as great a variety of molecules as carbon atoms can.Ĭarbon's ability to form long carbon-to-carbon chains is the first of five reasons that there can be so many different carbon compounds a molecule that differs by even one atom is, of course, a molecule of a different compound. 
They can even be shared by other carbon atoms, which in turn can share electrons with other carbon atoms and so on, forming long strings of carbon atoms, bonded to each other like links in a chain. The carbon atom's four valence electrons can be shared by other atoms that have electrons to share, thus forming covalent (shared-electron) bonds. Two are in a completed inner orbit, while the other four are valence electrons-outer electrons that are available for forming bonds with other atoms. Having the atomic number 6, every carbon atom has a total of six electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
